Concentrations of particulate nitrate (NO 3 − ) and nitric acid vapor... | Download Scientific Diagram

Concentrations of particulate nitrate (NO 3 − ) and nitric acid vapor... | Download Scientific Diagram
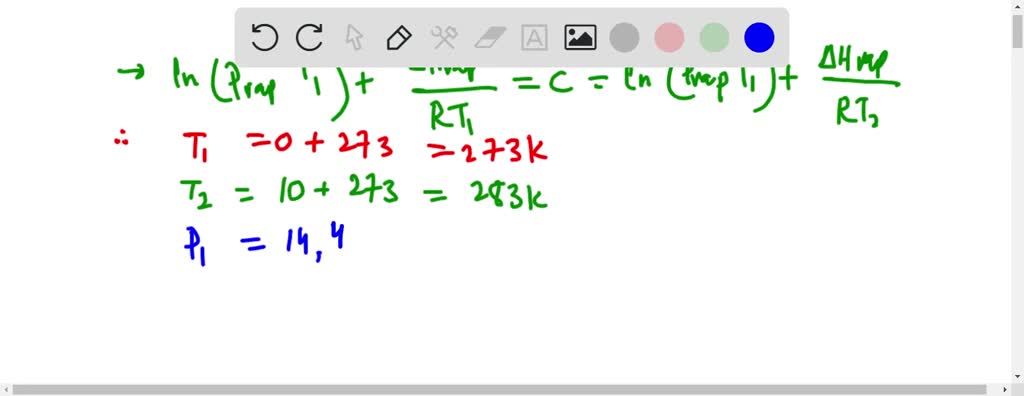
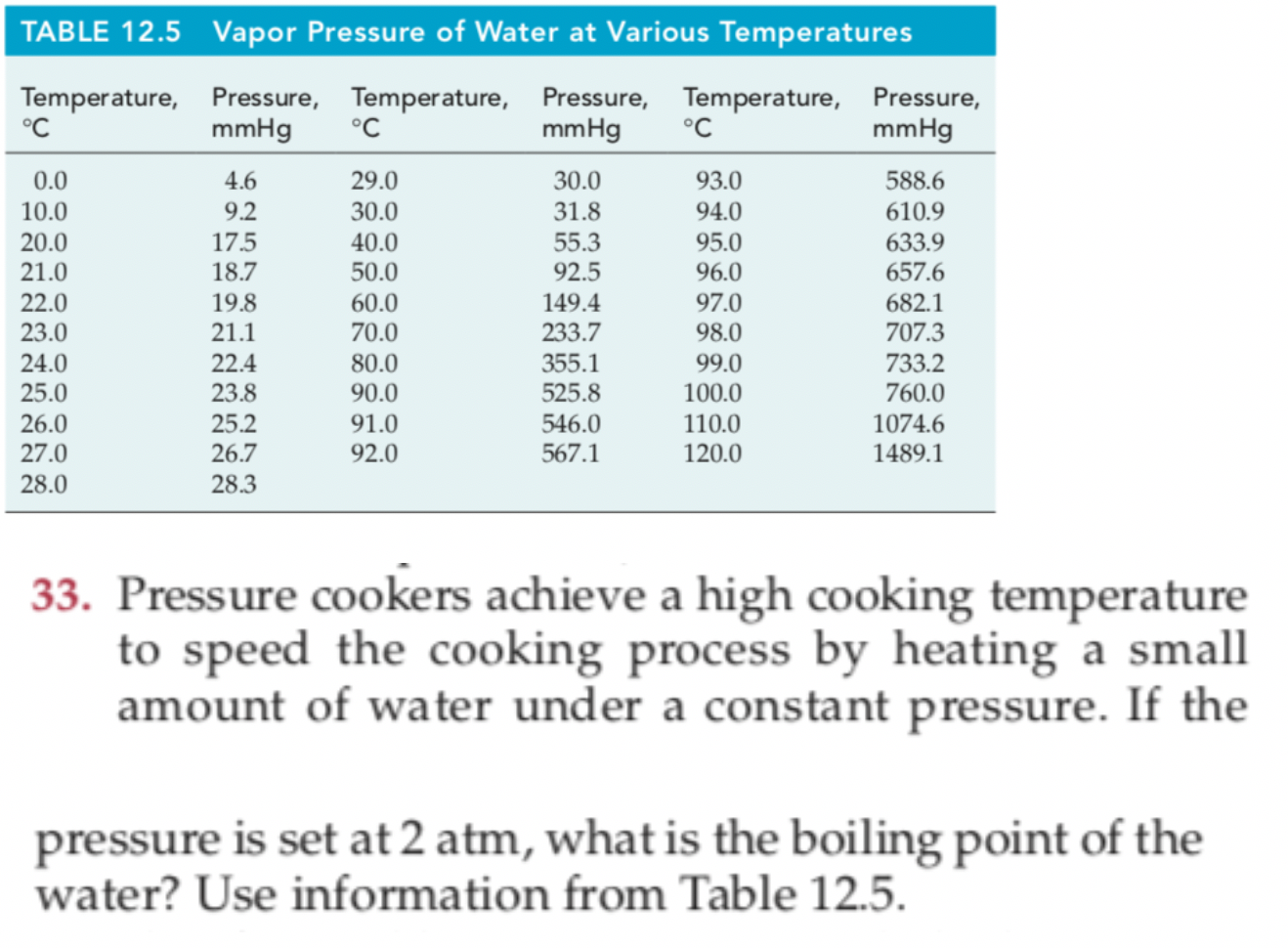
SOLVED:From the following data for liquid nitric acid, determine its heat of vaporization and normal boiling point. Temperature (^∘C) Vapor Pressure (mmHg) 0 14.4 10 . 26.6 20 . 47.9 30 . 81.3 40 . 133 50 . 208 80 . 670 .
Application of “Hydration Model” to Evaluate Gas Phase Transfer of Ruthenium and Technetium from Reprocessing Solutions

SOLVED:From the following data for liquid nitric acid, determine its heat of vaporization and normal boiling point. Temperature (^∘C) Vapor Pressure (mmHg) 0 14.4 10 . 26.6 20 . 47.9 30 . 81.3 40 . 133 50 . 208 80 . 670 .

Concentrations of particulate nitrate (NO 3 − ) and nitric acid vapor... | Download Scientific Diagram