CLARINET FORTE Baseline Characteristics: Lanreotide Autogel 120 Mg (LAN) Every 14 Days in Patients with Progressive Pancreat

Efficacy and safety of high-dose lanreotide autogel in patients with progressive pancreatic or midgut neuroendocrine tumours: CLARINET FORTE phase 2 study results - European Journal of Cancer
PDF) Novel Tumor Growth Rate Analysis in the Randomized CLARINET Study Establishes the Efficacy of Lanreotide Depot/Autogel 120 mg With Prolonged Administration in Indolent Neuroendocrine Tumors

Efficacy and safety of high-dose lanreotide autogel in patients with progressive pancreatic or midgut neuroendocrine tumours: CLARINET FORTE phase 2 study results - European Journal of Cancer
ESMO 2020: Phase II CLARINET FORTE results show increasing dose frequencies of Somatuline® Autogel® (lanreotide) allows patien
![PDF] Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study | Semantic Scholar PDF] Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a82b998becc9ea2f23aed89d6c17821920c6b73b/4-Figure1-1.png)
PDF] Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study | Semantic Scholar
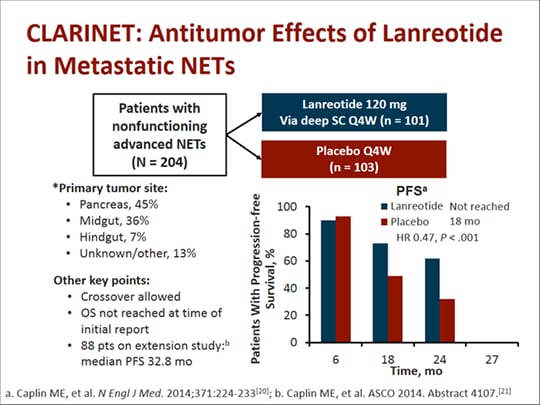
Essential Concepts in the Use of Somatostatin Analogues in Patients with Neuroendocrine Tumors (Transcript)
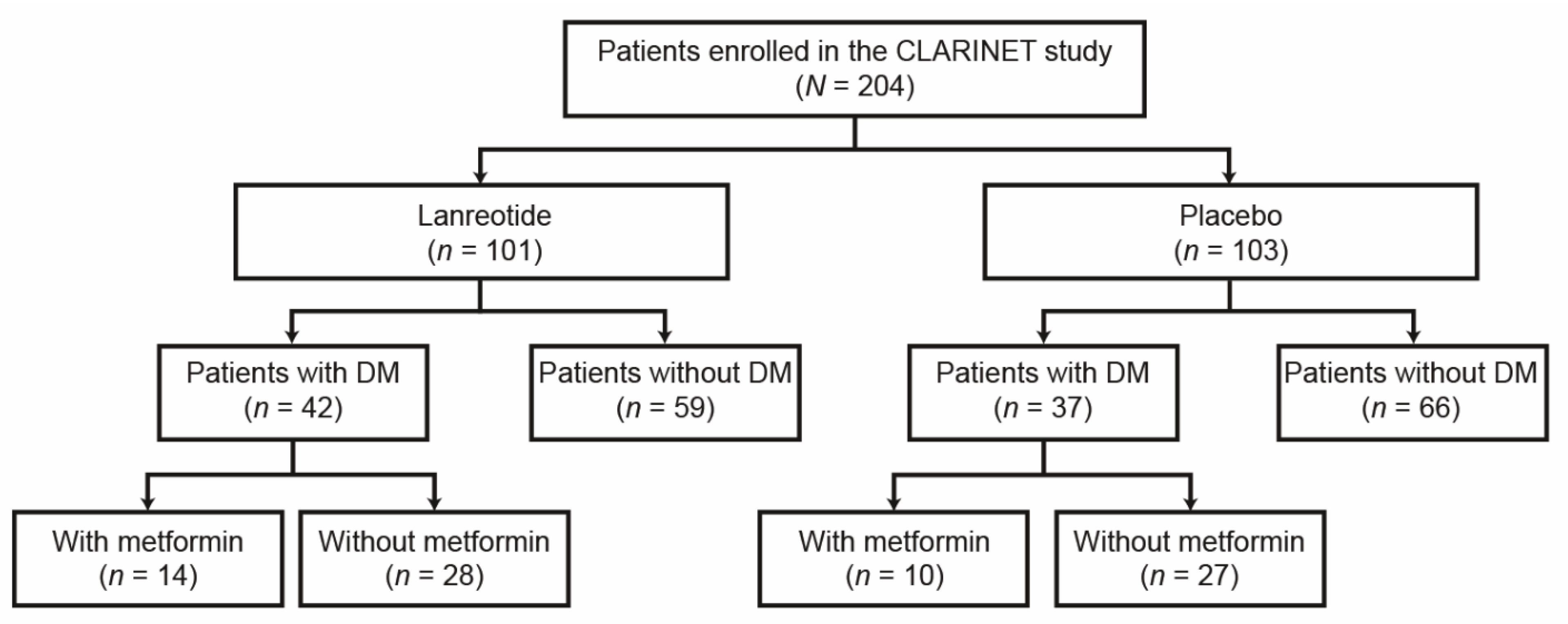
Cancers | Free Full-Text | Impact of Diabetes and Metformin Use on Enteropancreatic Neuroendocrine Tumors: Post Hoc Analysis of the CLARINET Study | HTML
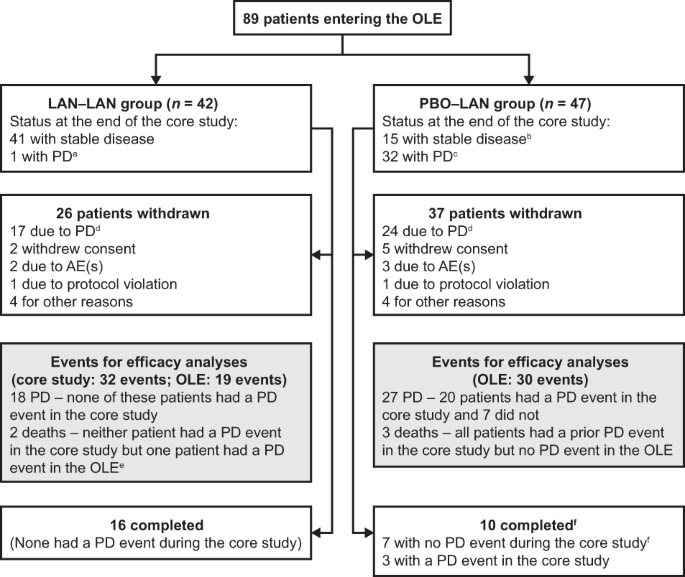
Lanreotide autogel/depot in advanced enteropancreatic neuroendocrine tumours: final results of the CLARINET open-label extension study | SpringerLink
![CLARINET FORTE study design [5]. ECOG PS Eastern Cooperative Oncology... | Download Scientific Diagram CLARINET FORTE study design [5]. ECOG PS Eastern Cooperative Oncology... | Download Scientific Diagram](https://www.researchgate.net/publication/360331878/figure/fig1/AS:1164524258365440@1654655627672/CLARINET-FORTE-study-design-5-ECOG-PS-Eastern-Cooperative-Oncology-Group-Performance.png)
CLARINET FORTE study design [5]. ECOG PS Eastern Cooperative Oncology... | Download Scientific Diagram
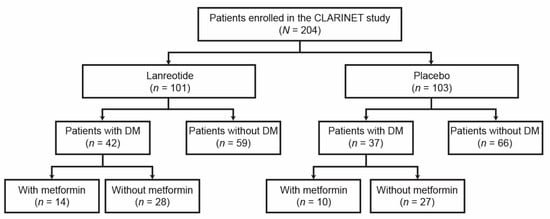
Cancers | Free Full-Text | Impact of Diabetes and Metformin Use on Enteropancreatic Neuroendocrine Tumors: Post Hoc Analysis of the CLARINET Study

Estimates of PFS among patients who received lanreotide depot (120 mg)... | Download Scientific Diagram
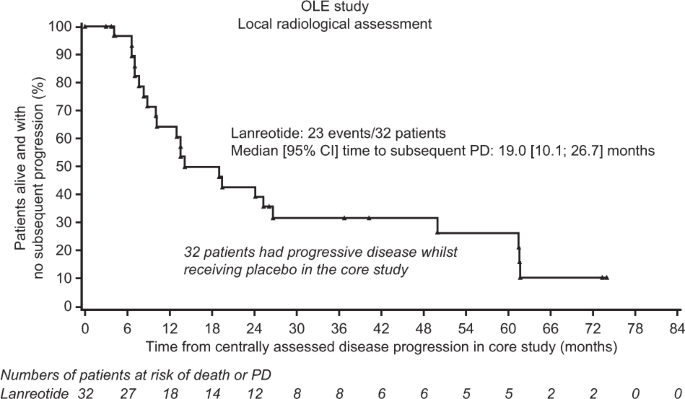
Lanreotide autogel/depot in advanced enteropancreatic neuroendocrine tumours: final results of the CLARINET open-label extension study | SpringerLink

Efficacy and safety of high-dose lanreotide autogel in patients with progressive pancreatic or midgut neuroendocrine tumours: CLARINET FORTE phase 2 study results - European Journal of Cancer
Duilio Rocha Filho on Twitter: "2⃣ CLARINET FORTE - lanreotide 120 mg q14 days in GEP-NET pts w/ progression on LAN 🤔PanNET: mPFS 5.6 mo 🤔Midgut: mPFS 8.3 mo 🚫Inferior results if
![PDF] Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study | Semantic Scholar PDF] Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a82b998becc9ea2f23aed89d6c17821920c6b73b/7-Figure2-1.png)
PDF] Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study | Semantic Scholar
![PDF] Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study | Semantic Scholar PDF] Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a82b998becc9ea2f23aed89d6c17821920c6b73b/6-Table2-1.png)
PDF] Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study | Semantic Scholar

Clinical Trial: Phase II CLARINET FORTE Somatuline Autogel (Lanreotide) increased dosing frequency - Ronny Allan - Living with Neuroendocrine Cancer
Lanreotide Autogel (LAN) 120 mg Every 14 Days in Progressive Midgut Neuroendocrine Tumors (NETs): CLARINET FORTE Study


