MIT Guide to PHS Human Subjects and Clinical Trial Information Form and Study Record for KC S2S and Workspace
MIT Guide to PHS Human Subjects and Clinical Trial Information Form and Study Record for KC S2S and Workspace

Efficacy and safety of hydroxychloroquine as pre-and post-exposure prophylaxis and treatment of COVID-19: A systematic review and meta-analysis of blinded, placebo-controlled, randomized clinical trials. - The Lancet Regional Health – Americas

Incorporating Site-less Clinical Trials Into Drug Development: A Framework for Action - Clinical Therapeutics
MIT Guide to PHS Human Subjects and Clinical Trial Information Form and Study Record for KC S2S and Workspace
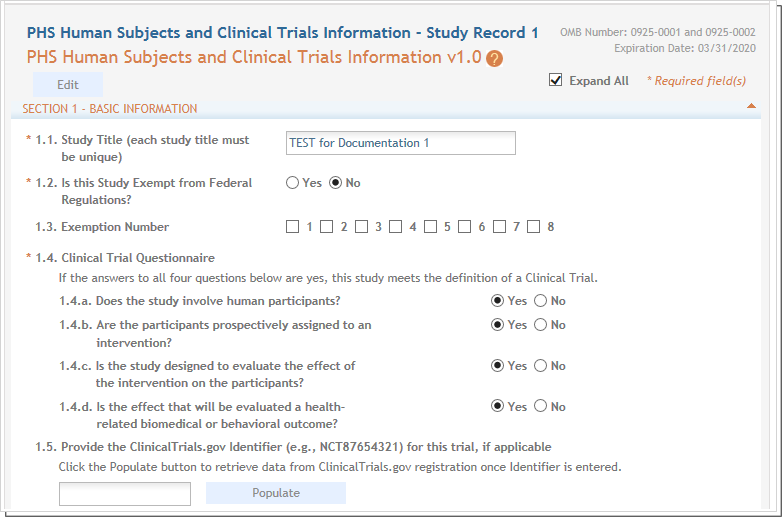
QUICK GUIDE PHS HUMAN SUBJECTS & CLINICAL TRIALS INFORMATION NIH HUMAN SUBJECT STUDY RECORD ATTACHMENT

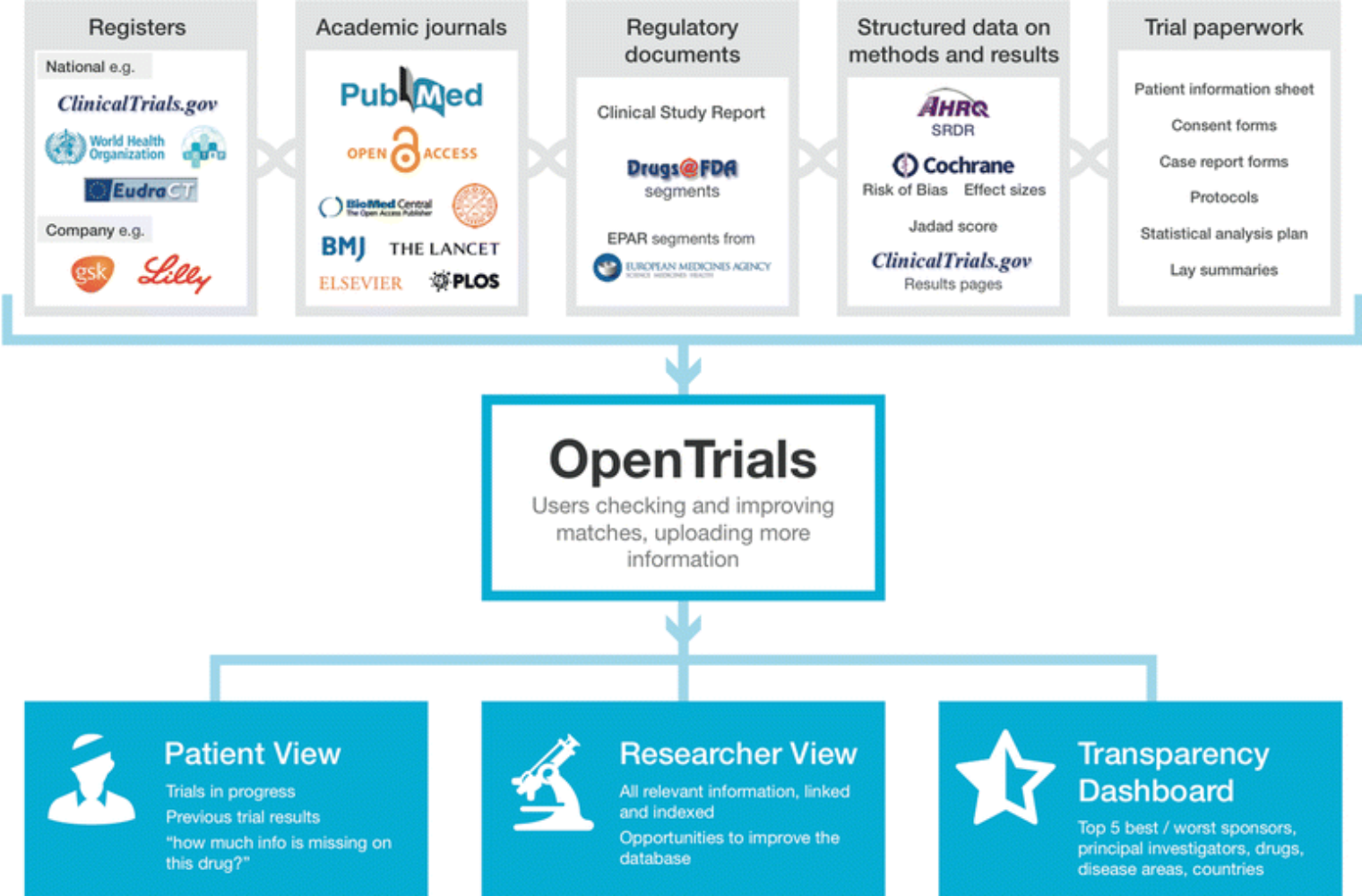
Celebrating 20 Years of ClinicalTrials.gov and Looking to the Future – NLM Musings from the Mezzanine

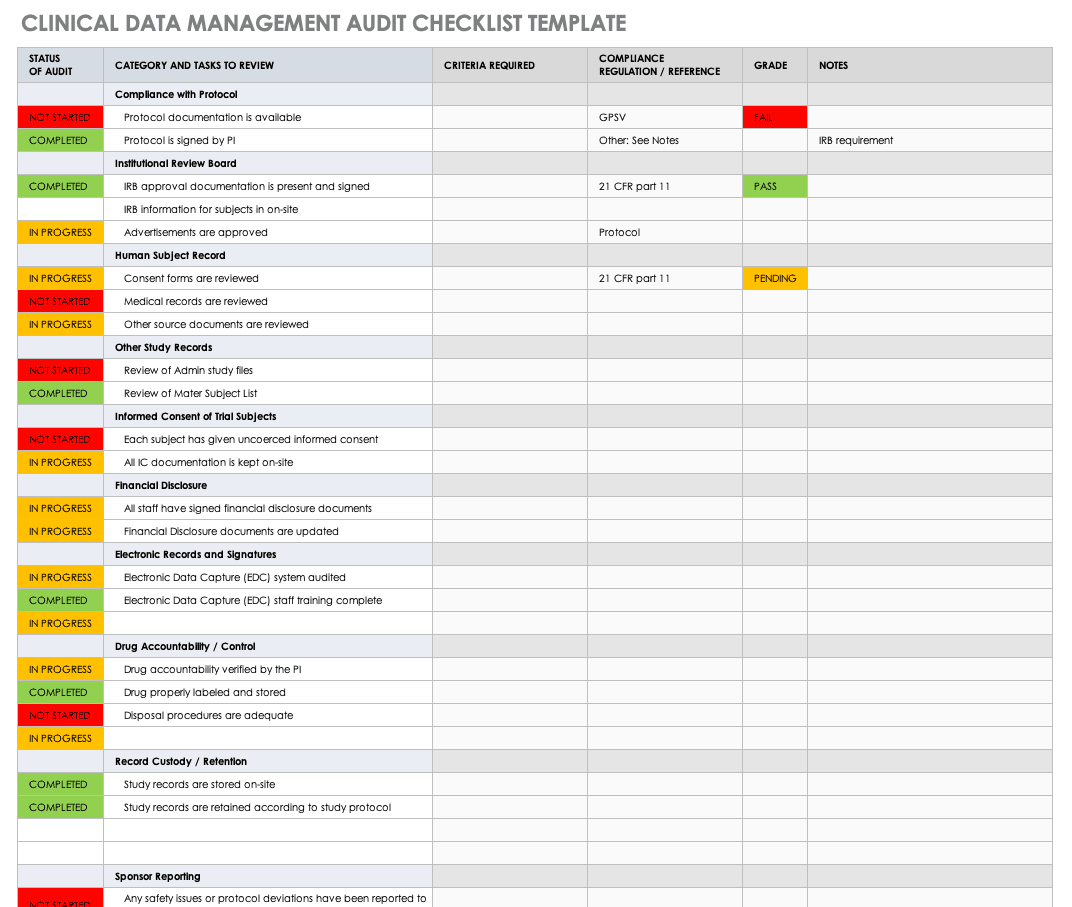
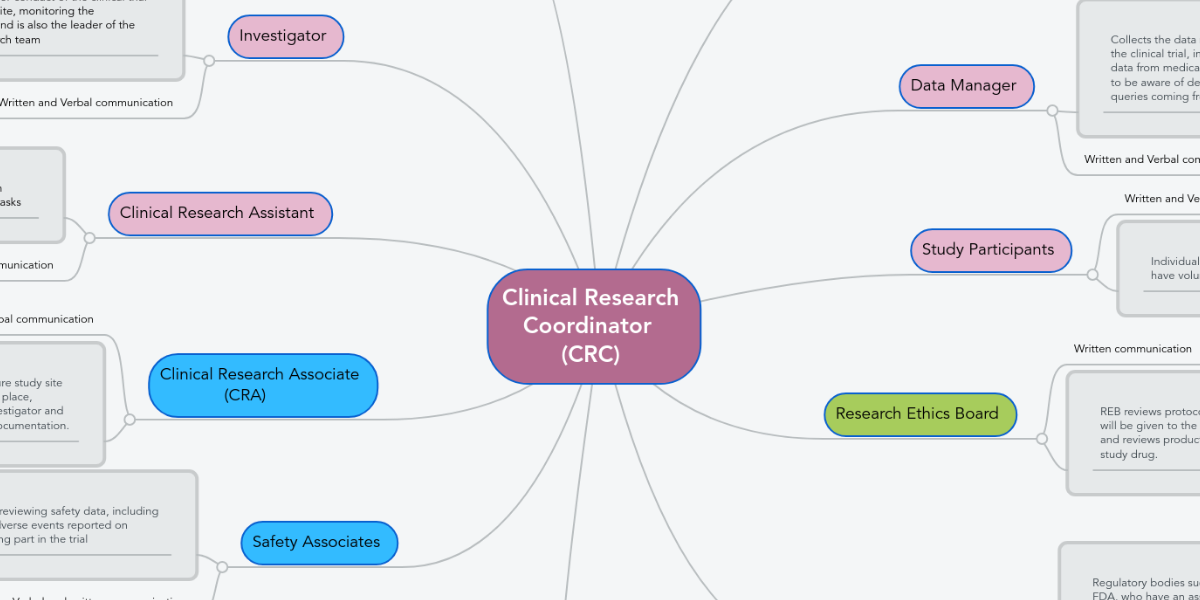
CT08: Clinical Trial Monitoring: Study Monitoring, Documentation and Closure | Zenosis – Learning for Life
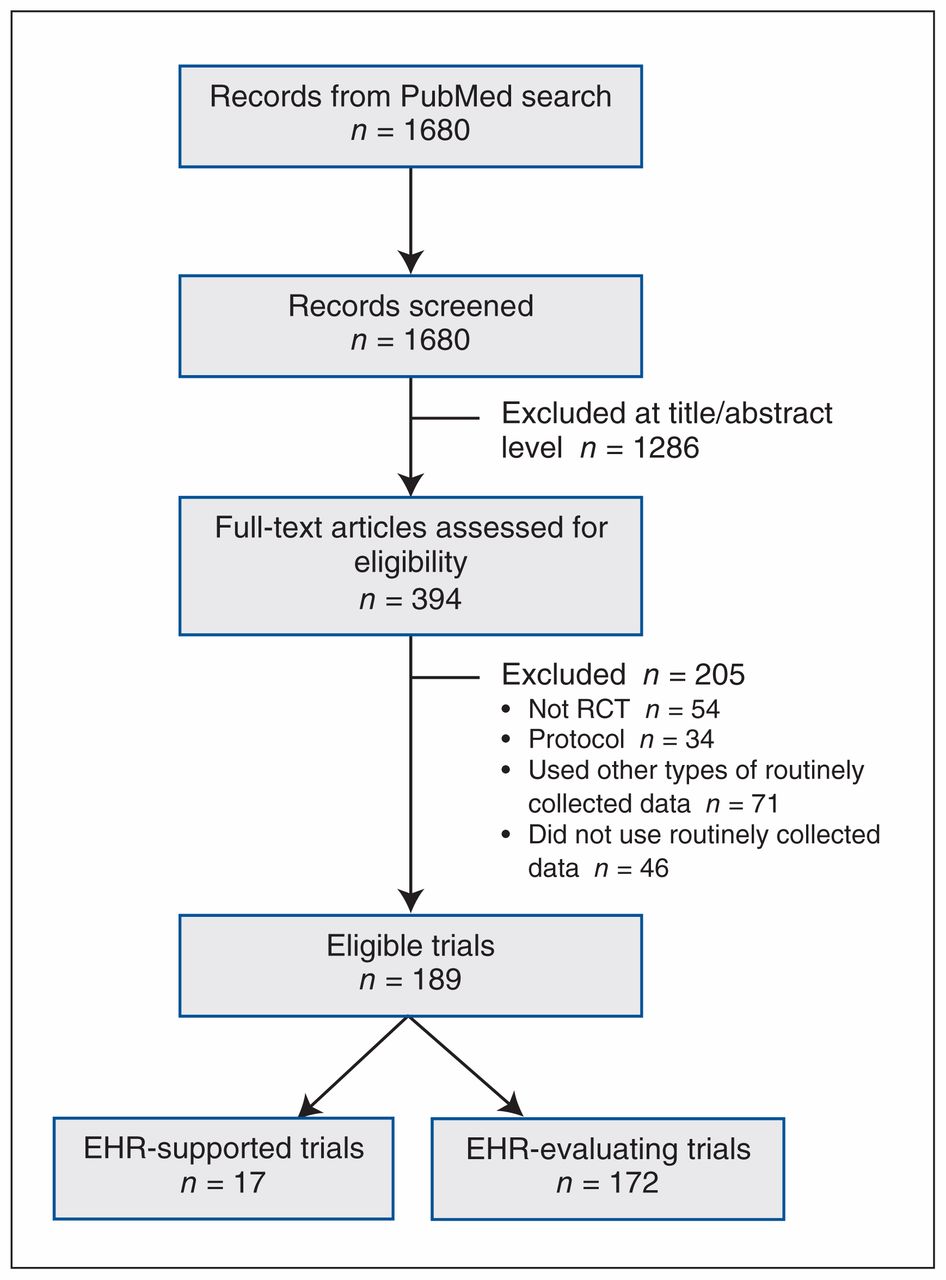
Current use and costs of electronic health records for clinical trial research: a descriptive study | CMAJ Open
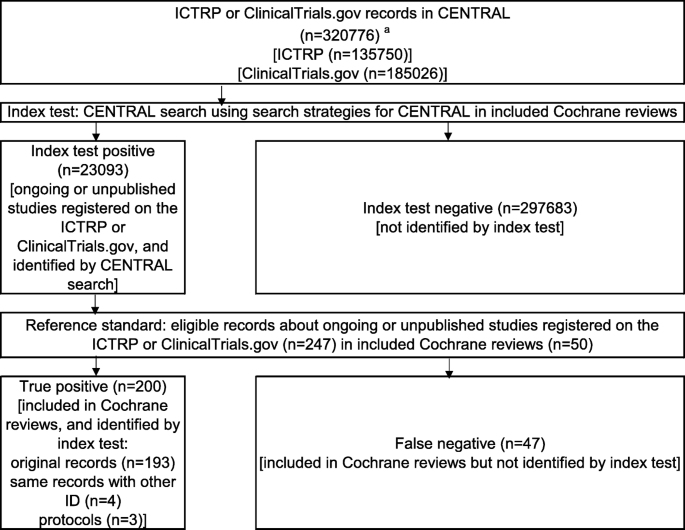
Using the Cochrane Central Register of Controlled Trials to identify clinical trial registration is insufficient: a cross-sectional study | BMC Medical Research Methodology | Full Text
Comparison between efficacy/safety and pragmatic trials. AE, adverse... | Download Scientific Diagram