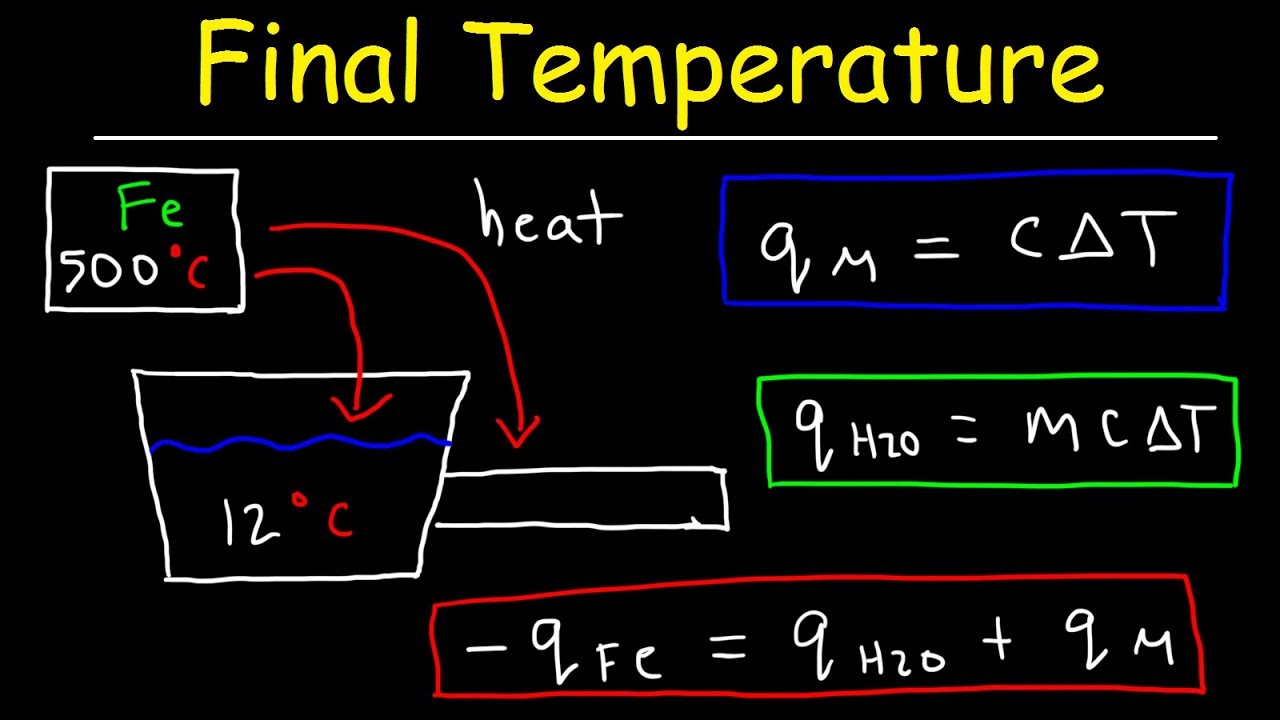
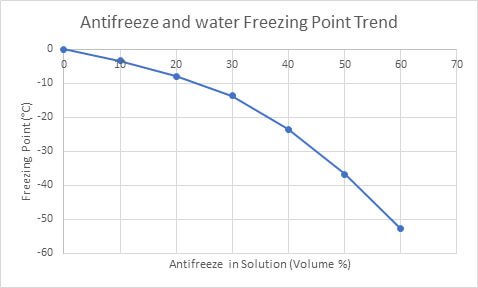
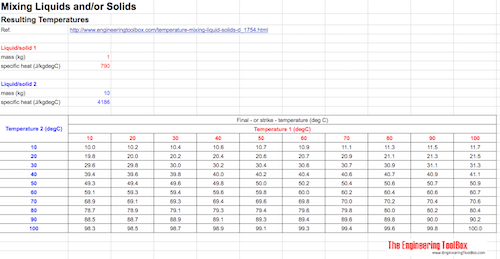
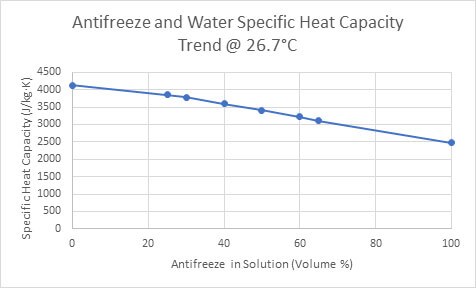
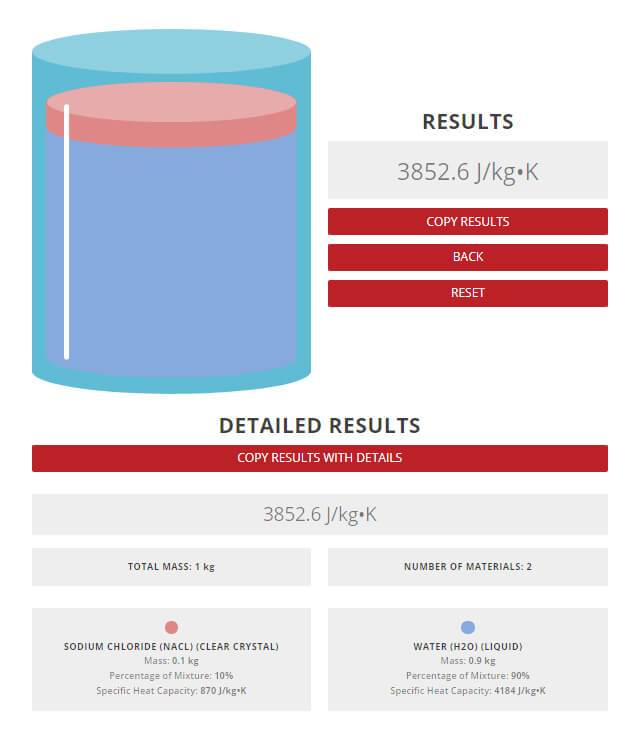
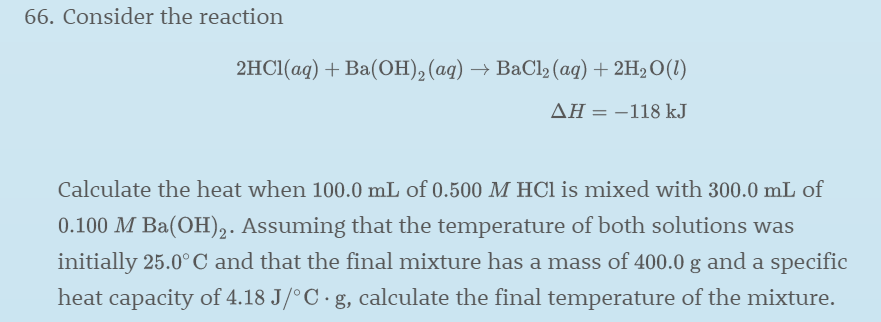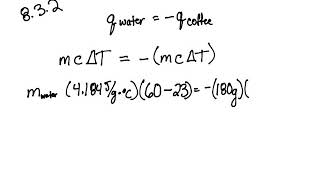SOLVED: Temperature before combining = 21.58C Time (seconds) 120 180 240 Temperature after combining( |23.5 Isc) 23.5 23.5 23.5 Calculate the energy absorbed by the calorimeter alone, Oclrinner Ocalorimeter = Xdelal =100X (

Two liquids X and Y form an ideal solution.The mixture has a vapour pressure of 400mm at 300K, when mixed in the molar ratio of 1:1 and a vapour pressure of 350mm

SOLVED:Consider the reaction 2 HCl(a q)+Ba(OH)2(a q) ⟶BaCl2(a q)+2 H2 O(l) ΔH=-118 kJ Calculate the heat when 100.0 mL of 0.500 M HCl is mixed with 300.0 mL of 0.100 M Ba(OH)2 .