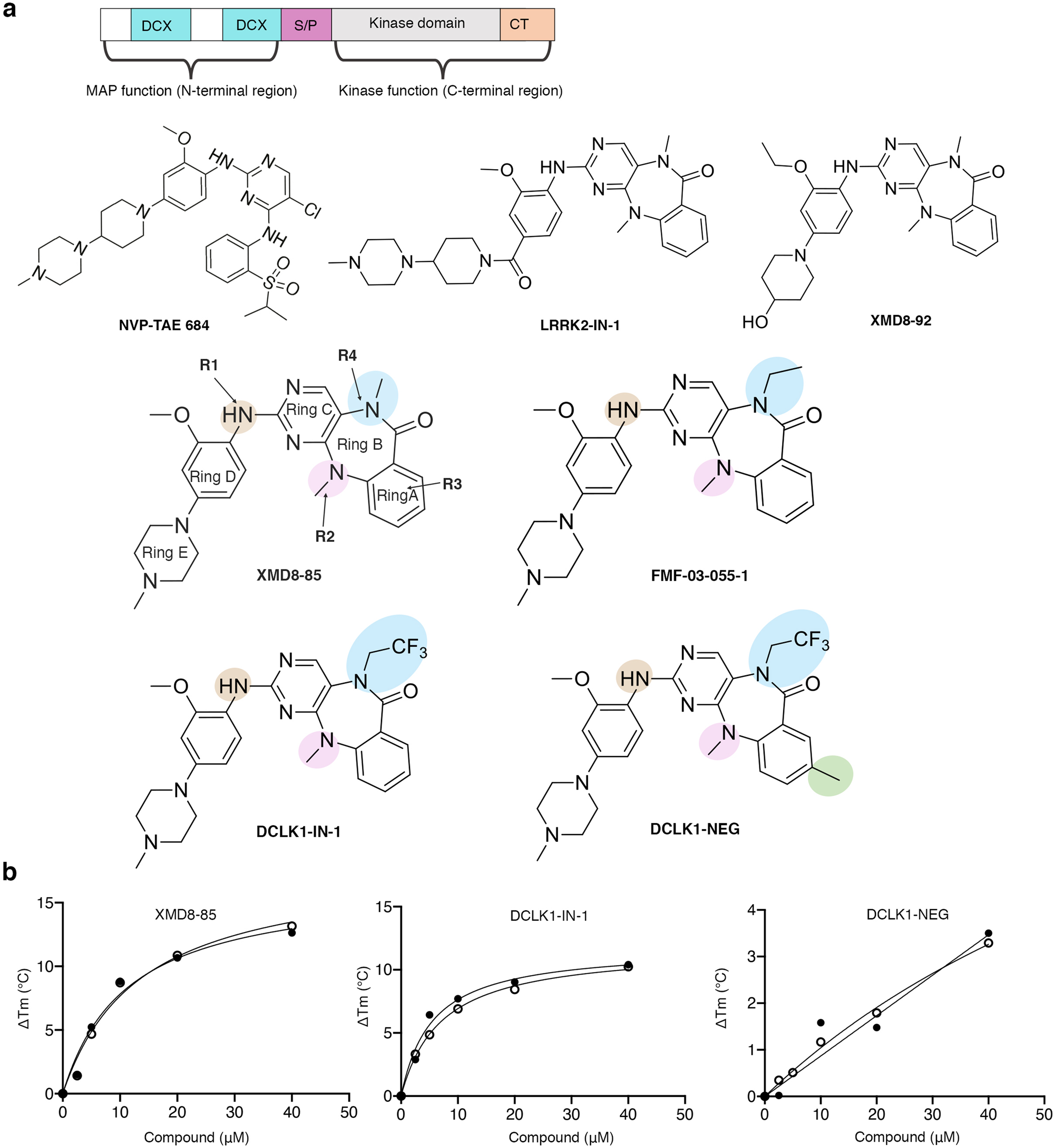
Structural basis for small molecule targeting of Doublecortin Like Kinase 1 with DCLK1-IN-1 | Communications Biology

LRRK2 regulates synaptogenesis and dopamine receptor activation through modulation of PKA activity. - Abstract - Europe PMC

Regulators of proteostasis are translationally repressed in fibroblasts from sporadic and LRRK2-G2019S Parkinson's patients | bioRxiv
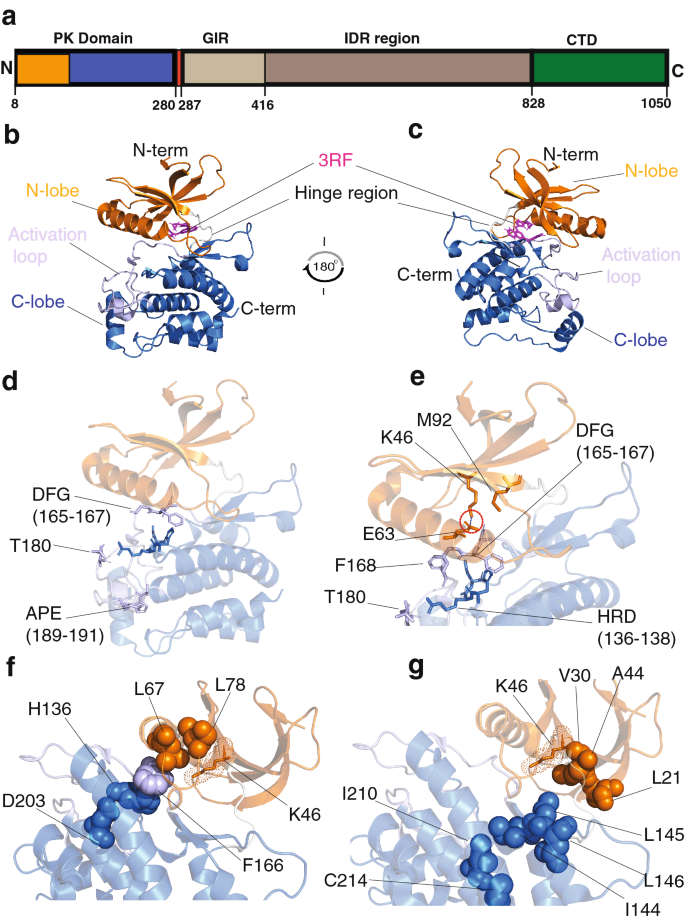
A pan-cancer assessment of alterations of the kinase domain of ULK1, an upstream regulator of autophagy | Scientific Reports
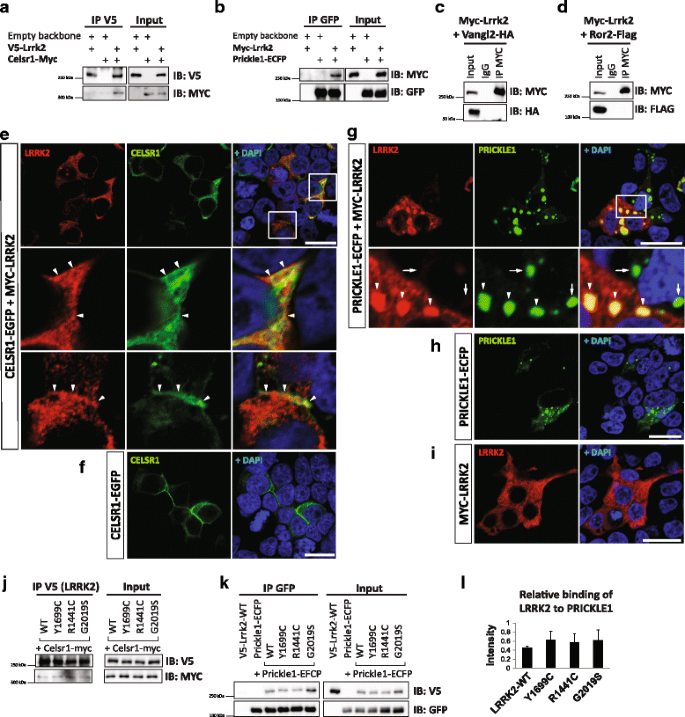
A proteomic analysis of LRRK2 binding partners reveals interactions with multiple signaling components of the WNT/PCP pathway | Molecular Neurodegeneration | Full Text
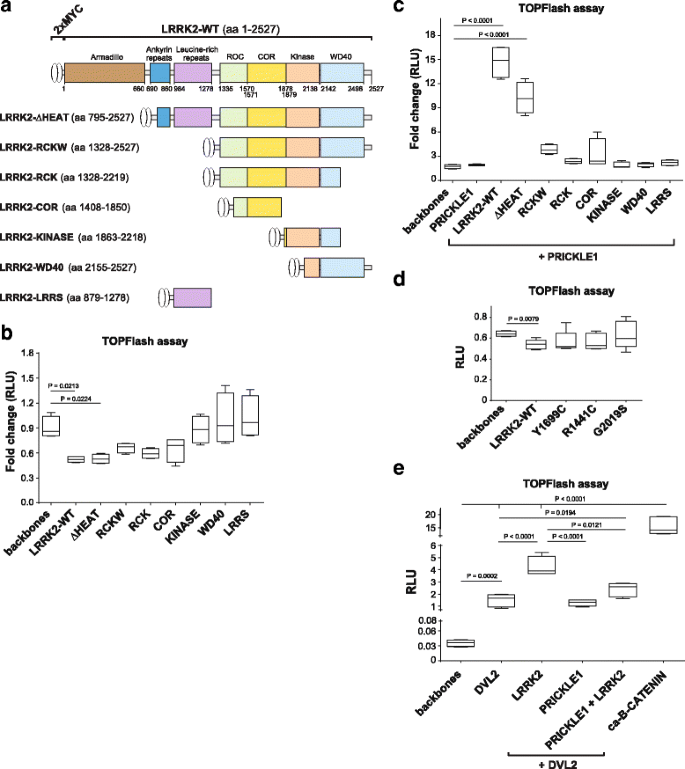
A proteomic analysis of LRRK2 binding partners reveals interactions with multiple signaling components of the WNT/PCP pathway | Molecular Neurodegeneration | Full Text

LRRK2 regulates endoplasmic reticulum-mitochondrial tethering through the PERK-mediated ubiquitination pathway. - Abstract - Europe PMC

Cocrystal structure of Roco4 kinase and compound 19 . (A) Overlay of... | Download Scientific Diagram

Leucine-rich repeat kinase 2 regulates the progression of neuropathology induced by Parkinson's-disease-related mutant alpha-synuclein. - Abstract - Europe PMC

LRRK2 G2019S mutation attenuates microglial motility by inhibiting focal adhesion kinase. - Abstract - Europe PMC
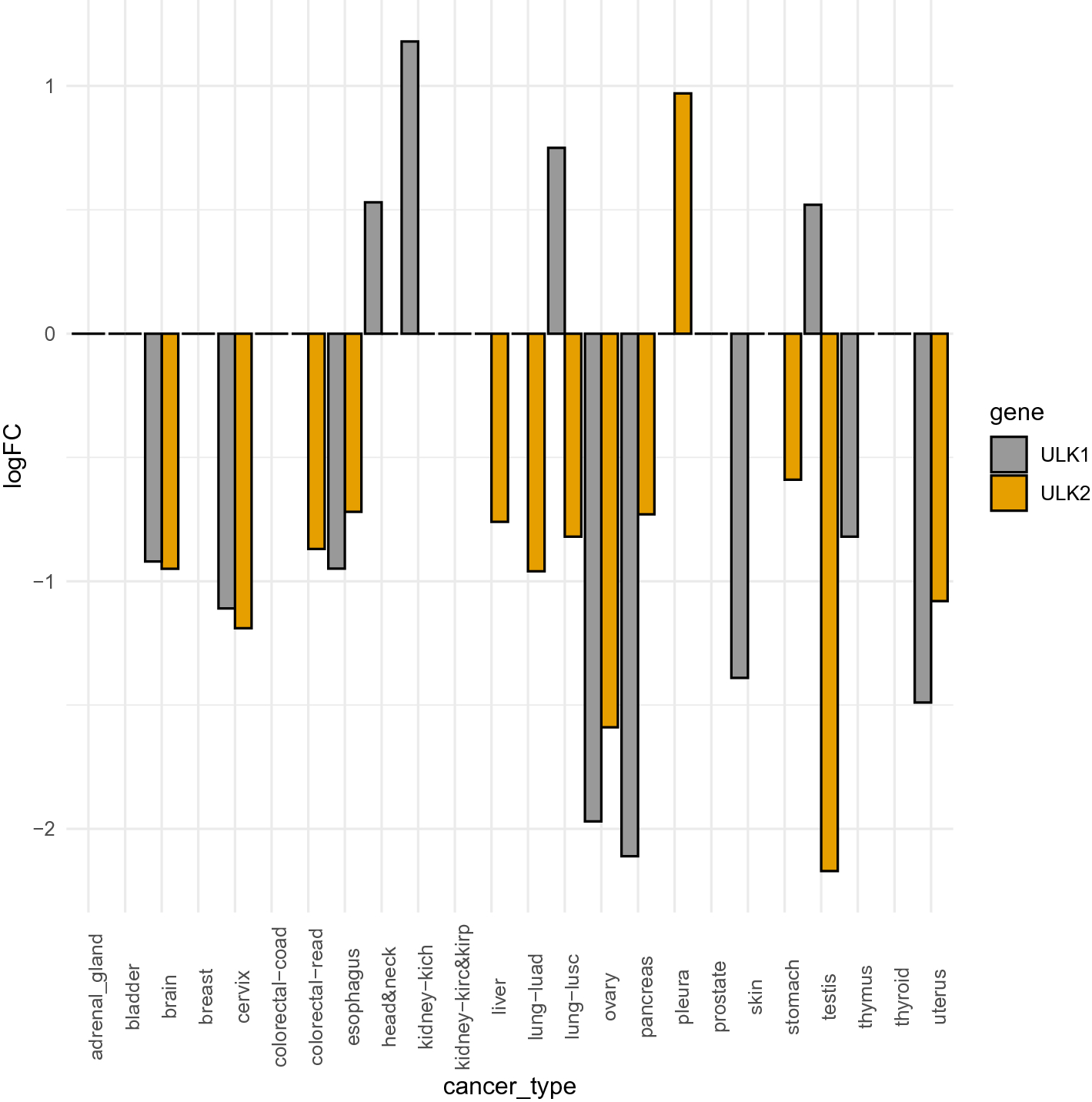
A pan-cancer assessment of alterations of the kinase domain of ULK1, an upstream regulator of autophagy | Scientific Reports

LRRK2 regulates synaptogenesis and dopamine receptor activation through modulation of PKA activity. - Abstract - Europe PMC







![Recombinant Anti-LRRK2 antibody [UDD3 30(12)] KO Tested (ab133518) | Abcam Recombinant Anti-LRRK2 antibody [UDD3 30(12)] KO Tested (ab133518) | Abcam](https://www.abcam.com/ps/products/133/ab133518/Images/ab133518-270971-anti-lrrk2-antibody-udd3-3012-western-blot.jpg)