Raport de activitate al Agenţiei Naţionale a Medicamentului şi a Dispozitivelor Medicale pentru anul 2017 Introducere Agenţi
REZUMATUL CARACTERISTICILOR PRODUSULUI 1. DENUMIREA COMERCIALĂ A MEDICAMENTULUI Borenar 2,5 mg/ml soluţie orală 2. COMPOZIŢ

PDF) Evaluation of the Single-dose Pharmacokinetics of Bilastine in Subjects with Various Degrees of Renal Insufficiency
Raport de activitate al Agenţiei Naţionale a Medicamentului şi a Dispozitivelor Medicale pentru anul 2017 Introducere Agenţi
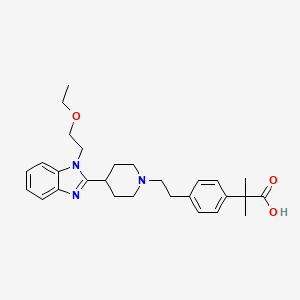
The role of bilastine in treating paediatric patients with allergic rhinoconjunctivitis and urticaria